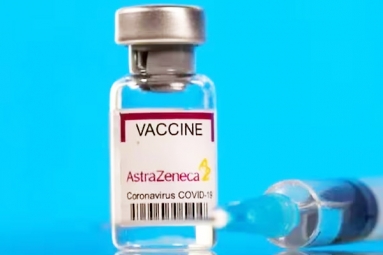
AstraZeneca's COVID-19 Vaccine Study on Hold as Serious Side-Effects Emerge
September 09, 2020 16:47
(Image source from: m.hindustantimes.com)
A participant in the UK has developed adverse side effects after being administered with AstraZeneca's vaccine shots.
The phase 3 study testing the COVID-19 vaccine being developed by Oxford University and AstraZeneca is being carried out in the UK and US.
The study is now being put on hold as a volunteer developed a serious reaction after taking the vaccine.
A spokesperson of the AstraZeneca said in a statement that the company's standard review process has been paused to review the safety data of the vaccine.
In a follow up statement, the company said that it is examining the nature of the adverse reaction.
The participant is expected to recover within a few days.
The spokesperson described the pause as a routine action which had to happen whenever there is any unexplained illness in the routine of the trials.
The pause is to ensure the company maintains the integrity of the trials.
The company is also working on expediting the review so that the timeline of the trial completion is not disturbed.
An individual who is aware of the development said that the researchers have told that the pause in the trials is to ensure safety and comes as a caution.
A second person who is also familiar with the process also said that the recent finding is having an impact on the vaccine trials of the company and also the trials conducted by the other manufacturers.
However, the holds on the clinical trials are not uncommon but it is not sure how long the hold on the trials of AstraZeneca will last.
There are currently 9 vaccine candidates in the phase 3 trials. AstraZeneca's is the first phase 3 COVID-19 trial known to have been put on hold.
Researchers running other trials are now looking for the similar cases of adverse reactions by combining through the databases reviewed by the Data Safety and Monitoring Board.
AstraZeneca had begun its phase 3 human trial in the US in late August. The US trial is currently taking place at 62 sites across the country, though some of them have not yet registered the participants.
Phase 2 and 3 trials were previously started in the UK, Brazil and South Africa.
There are a number of different reactions that can qualify as suspected serious adverse reactions, and some of the symptoms need hospitalization, life threatening illnesses and even deaths.
However, it is still unclear how severe and rare the adverse event may be.
- By GayatriYellayi